A COLORIMETRIC METHOD FOR THE QUANTITATIVE
DETERMINATION OF NITRATES AND NITRITES
IN BIOLOGIC FLUIDS- http://www.jbc.org/content/86/1/189.full.pdf
I don’t have access to a lab with colorimeter. @jaikishan is there a colorimeter available at CUBE Lab that could be used. Another problem is I would have to monitor nitrates daily and I can’t go daily at CUBE Lab.
But even if I could we need some method that could be easily done by anyone because if we are going to build a model then a few number of data points aren’t gonna give accurate information.
Something like titration will work since all of us can do it some way or the other and even the most basic chemistry labs are well equipped for this task.
Any other suggestions? Or should we completely throw away the idea of monitoring nitrates and go in some other direction to quantitatively measure the changes in waste amounts?
How can the condition in moina bottles be oxidized when bacteria are increasing day by day and consuming up all oxygen?
Not clear what do you mean by polar?
Why not exactly tell what is needed for estimating nitrates and nitrites with titration method?
What is the principle involved?
What are steps involved?
most of these assays can be done in any college labs…
Please describe in detail steps involved in this… @pratittodkar
Qualitative assays can also be done inorder to know which chemicals are formed as days pass in moina bottles.
I hope you have qualitative and semi qualitative assays in your practical syllabus of chemistry.
Please describe how it can be helpful in this challenge… @pratittodkar
- The oxygen is being consumed that doesn’t mean that it isn’t there. It just means that it is combined with some other chemicals like carbon or any other element to form there oxides.
Example: When it combines with carbon to form carbon dioxide
Hence, condition in Moina bottles gets oxidized.
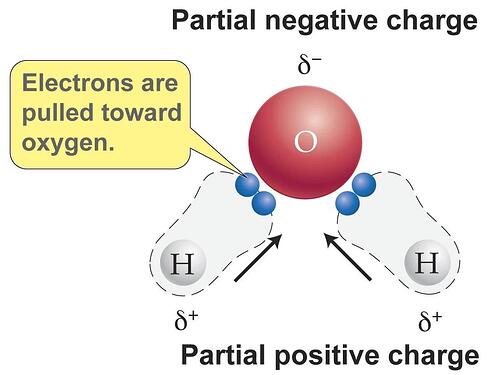
- When formation of covalent bonds take place they between atoms of elements with unequal electronegativity then the bond formed is non symmetrical in charge. Meaning one atom gets a partial negative charge and the other one gets a partial positive charge. This forms two poles in the molecule +ve and -ve. Hence they are called polar molecules.
-
The problem is that I exactly haven’t found a way for titration of nitrates and nitrites. I don’t know how to carry out titration for nitrates or nitrites. That’s why I have been asking for ways to quantitative tests for them.
-
There is a way to do potentiometric titration but it just seems like an overkill.
-
Instead we can just do titration for ammonia and when we detect a reduction in ammonia levels or a reduction in ammonia growth rate, it will be due to ammonia oxidizing bacteria. As it would be the only source consuming ammonia to form nitrites. Hence can estimate how much of ammonia is consumed.
-
Though this method can give false positives when ammonia forming factors change, it seems very unlikely that they will.
-
@jaikishan I would like to know your opinion on this subject. I am quite naive at chemistry so if you have any interesting insight about this matter I would love to try it out.
- These I am gonna do but qualitative analysis won’t be sufficient we will also have to do quantitative analysis.
- Qualitative assays will be only useful in detection not in estimation of quantity. That’s the problem qualitative analysis isn’t that useful in this case since we are mainly concerned with quantity. Though we sure will do detection but that can easily be done hence the main concern is about estimation.
About pH titration of ammonia using HCL.
I also want a opinion about how to estimate the number of bacteria in Moina bottles? Should we just look for a particular bacteria or should we look for any kind of bacteria we can find?
Till now, I didn’t find any way to estimate the number of bacteria but we can look at the turbidity in the bottles. More turbidity, more bacteria.
It is difficult to look for a particular bacteria. From where will you obtain any kind of bacteria?
Even if obtained, the feeding needs to be done in sterile conditions!!
I am with you on the problems for estimating bacteria but I don’t think that turbidity is the way to go since the water is going to get murkier even without bacteria because of milk particulates. The oils and fats from milk will give more chances of false results. I don’t know the correct way to go about this problem but I don’t think turbidity will work. @drishtantmkawale any other suggestions?
@pratittodkar isnt moina in bottles turn red precisely because oxygen is used up and not available?
How can bottle of moina be oxidized when oxygen is used up and other reduced products are formed.
Is there any oxidized chemicals which smell? or those which have smell are reduced compounds?
Please give some examples of chemicals which smell, are they oxidized or reduced compounds?
From where will ammonia come in moina bottles? will it increase as time passes or will it decrease?
Why not list which qualitative assays you are aware of?
Ammonia is basic in nature and can be titrated with acid, to give salt and water, which can be done in any chemistry lab, but as we dont have pure ammonia, so this method cant be used.
http://simulab.ltt.com.au/4/laboratory/methodsman/MMSOP-DetmntnAmmoTitSulfAcid.htm
Why not look for Nessler`s reagent method inorder to determine ammonia?
@drishtantmkawale why not try estimating bacterial count from moina bottles as days pass?
why not use some simpler techniques like hemocytometer to find approximate count under microscope? This can be done in an average college.
Ya they turn red due to absence of pure oxygen. But not due to absence of oxygen compounds. There are oxygen compounds present which are formed when other elements are oxidized as they react with oxygen.
As I said smell isn’t associated with oxidation or reduction. Instead it is associated with the polarity of the compound. And oxidation and reduction don’t determine polarity instead it is determined by the electronegativities of elements in the compound.
There are many compounds which contain oxygen and still have odor and other which do not contain oxygen and don’t have odor.
Example: Benzene (Doesn't contain oxygen) Benzyl acetate (Contains Oxygen)
Ammonifying bacteria make ammonia from decomposing organic matter. It will increase as time passes.
Lets look at how we can measure ammonia? How it is done in college practical syllabus? @pratittodkar @drishtantmkawale
Yes!!
Bacillus, Streptomyces, Clostridium as indeed ammonifying bacteria.
And as the bacteria will increase, NH3 will also increase.
Determination of Ammonia (a base) can be done by titrating it with H2SO4 (an acid).
Reference: http://simulab.ltt.com.au/4/laboratory/methodsman/MMSOP-DetmntnAmmoTitSulfAcid.htm
We don’t have that in our college syllabus. We only have titration for Oxalic acid and KMnO4
- NH4+
- N
- S
- Cl
- Br
- I
- Cu2+
- Al3+
- Ba2+
- Ni2+
Oxidation means the addition of oxygen to a molecule or the removal of hydrogen from a molecule. Reduction means the addition of hydrogen to a molecule or the removal of oxygen from a molecule.
Which means electronegativity is directly related with oxidation & reduction.
Benzene smells because it is reduced compound. So reduced compounds do have smell. Benezene is further reduced to cyclohexane which also has detergent like smell.
Also those compounds which dont have carbon may have smell, like H2S, CH4, NH4, etc.
It will be great if we understand nature of molecules which have smell and which dont have smell…