[size=5][center]Let Us Know Our Fruit Flies[/center][center](A Basic Guide to the Six-times Nobel Prize-Winning Flies)[/center][/size]
Our Collaborators:
Aashutosh Mule, Abhijith Vinod, Abhishek Unnikrishnan, Anna Malai, Ankit Yadav, Apoorva Satpute, Arunan MC, Aswathy Suresh, Batul Pipewala, Deepika Iyyangar, Drishtant Maruti Kawale, Ganesh Chandra Baskey, Harshita Bhanushali, Hina Mudgal, Isha Pawle, Kavya Honnavad, Kiran Yadav, Kshipra Biresh, Lydia Mathew, Man Masih Beck, Mayur Gaikwad, Nagarjuna G, P. Chitralekha, Rahul Kushwaha, Ruchi Modgekar, Saida Sayyed, Savithri Singh, Shalu Sinha, Sheetal Rana, Sidhy PP, Yamal Gupta, Yash Sheregare.
1. Introduction
[center]FRUIT FLIES[/center]
[floatl]
[/floatl]Ever heard of them? Many of us must have heard of it in the science class or from textbooks. But what if we ask a watchman, a gardener, or an uncle/aunt you meet during morning/evening walks. How will you explain or tell them what a fruit fly is? Let’s try to make them understand in simple words what it is and why is it important to know about it!
Fly in the Kitchen: These are one among the many types of small flies that we often see in our kitchens. You find them buzzing around over your fruit basket or any place where you keep your fruits like mango, banana, etc. It can also be seen over rotting, rotten fruits or vegetables and especially near their peels which we throw in the dustbin.
This fly is what we are talking about.
It is the fruit fly!
The common fruit fly, also known as the vinegar fly, due to the fact that it is attracted to vinegar-like smell produced by over-ripe fruits, is scientifically known as Drosophila (Drosophila melanogaster is one of the species of the Family Drosophilidae)
And why is this fly special? It is because of the fact that it has proven to be a useful model system to understand many biological phenomena. Thus, various studies using these flies have turned out to become pathbreaking, read Nobel prize-winning work. As many as 6 Nobel prizes have been awarded until now , to scientists for their discoveries based on experiments carried out on this fly. Thus, it is special.
Let’s go on to know more about this ‘Nobel’ organism through a series of exercises and experiments that can be done by all of us with ease and with few inexpensive instruments.
What is a fruit fly called in our local language?
An interesting part of this exercise will be to start with knowing the different names of the Fruitfly in different regions, in different local languages. Many times, the fruit fly may not have a separate name; all small flies are together known by a common name!
So, when we asked people belonging to different states, we got different answers-
[Here is a story by Shalu from Patna, Bihar to begin with,
“When I did not have bananas or tomatoes, I used to ask people here if they have, and they used to ask me the reason, and they were keen in listening to me, but when I would say ‘drosophila’, they did not understand and felt very alienated about the organism.
But my mother once told a relative,- ‘She wants to catch pakhni, for her study’
Then I was enlightened by responses such as, “ooh pakhni, they are attracted to muskmelon also. You can use it if bananas and tomatoes are not available.”]
So, we went about compiling the names for fruit flies from different states of India.
The local names of the fruit flies in different languages/dialects:
- Uttar Pradesh: Bhunge, Maachi, Choti Makkhi
- Haryana: Choti Makkhi
- Bihar: Pakhni
- West Bengal: Phaler Maachi
- Kerala: Podi eecha, Kuzhi eecha (Thrissur), Kunjan eecha (Ottapalam, Palakkad), Phalam eecha, Ponni eecha (Mallapuram), Kuyi eecha (Pathanamthitta), Payam eecha, Manian (Edappal), Kuruteecha (Thiruvananthapuram)
- Maharashtra: Mashi (a common name for all the flies visible around, for example, Fruit flies, house flies), Chilte, Kemra, Chachan, Murkut in (Kolhapur).
- North Goa: Muskan (muskan with ‘n’ silent, mus pronounced as moose is house fly)
- Jharkhand: The Santhali Tribe calls it Rhon and the Oraon Tribe calls it Tingli the place of the Oraon tribe is Urukh and their dialect being Kudukh. Rhon and Tingli is the general name of any fly. Machchi in Jharkhandi, Sadri dialect.
- Rajasthan: Kutaar in Marwadi.
- Odisha: Machhi.
- Gujarat: Maskidi.
- Karnataka: Hannu nona, Gungra (common name for all small flies) in Bangalore and Domari in North Karnataka.
- Tamil Nadu: Pala ee.
- Uttarakhand: In Garhwali, a native language of the Garhwal district, the fruit flies are called Jhiju (pronounced as Jhi-ju).
- Assam: Kunkuni Makkhi where Kunkuni means small in Assamese and Thamfwi ongkha in Bodo language.
- Manipur: Ponua in Bishnupriya Manipuri and Juran in Meitei Manipuri languages.
- Dubai/ Abu Dhabi: Zubabatul fakehate the Arabic language.
- Telugu: Phala eega, Pandu eega (Phala/Pandu= fruit, eega=fly)
2. HOW TO LOCATE AND TRAP THE NATIVE FRUIT FLIES IN OUR NEIGHBOURHOOD?
(Topic: Field biology)
For trapping Fruit flies -
[floatl]
[/floatl]Materials required:
1.Any ripe fruit, but most preferably fruits that are available throughout the year (slices of ripe tomato, banana, lemon being easily available in our houses) can be baits to attract flies. (Note: Let’s also try out some raw fruits, cut onion, pieces of cucumber etc.)
2. Transparent bottle or container (plastic or glass bottle/jars)
3. Cotton plug, cotton cloth piece or newspaper (to cover the mouth of the bottle/jar once the flies enter it)
Procedure -
We can set up two types of traps:
[center] [/center]
[/center]
1 - An open bait.
- In an open bait set up, we simply keep the bait of our choice on a paper sheet/plate/bowl near the kitchen window/bin, in order to attract the fruit flies.
2 - A bottle/bucket trap with bait inside.
- We can keep the bait (pieces of banana/tomato or even peels of fruits) in a bottle and wait for the fruit flies to come.
- After introducing the bait in the bottle, the mouth of the bottle should be cleaned with a piece of paper or clean cotton so that when the flies come, they may not roam around the neck (as there is a chance that some part of the fruit might have brushed along the neck due to which particle of fruits may stick on the neck too), but go down inside the bottle.
[center]
 [/center]
[/center]
- Once we get flies inside, we can close the mouth of the bottle with newspaper sheets or tissue paper with tiny holes, or cotton plugs.
Queries:
What will happen to the flies trapped inside the bottles?
a. Would they be alive after a day or two? How about after a week?
b. Would they feed, mate & multiply?
c. Would the flies behave as if it is their natural environment? What is the natural environment of the fruit fly in the wild?
3. DO FRUIT FLIES SLEEP?
Understanding Sleep-Wake Cycle, if any, by Documenting Daily Activity Pattern of Fruit Flies.
Life on Earth is strongly influenced by the daily cycles of light and darkness imposed due to the rotation of earth on its own axis. As a consequence, living systems have evolved internal timing mechanisms that help them to anticipate these daily changes in their environment such as gradually rising and falling light levels and temperature, availability of food, predators, or mates at only specific times of the day / night. These internal clocks are referred to as ‘circadian’ clocks from Latin, Circa - approximately, Dies - a day. As humans, we are made aware of our internal, daily or circadian clock when we find ourselves waking before the alarm rings each morning, or tend to feel sleepy at our regular bedtimes at night, or feel the uncomfortable repercussions of late nights in the form of low alertness or lack of general well-being the next day.
But why should we study the daily rhythms of Fruit flies?
We may think that Circadian Rhythms occur in humans too, then why study this behaviour in fruit flies. We are studying it in fruit flies because they can be easily trapped, they can be maintained at home-labs and in school/college-labs (in large numbers) and also, their life cycle gets completed in 10-12 days. Moreover, these flies exhibit daily rhythms in several behaviours including patterns of their activity-rest, feeding, mating, egg laying, propensity to emerge out of their pupal cases (eclosion), etc., AND, THEY ARE THE NOBEL-PRIZE WINNERS…(see below)!
When we keep a rotten fruit or vegetable and observe it closely after some time, we will be able to observe flies wandering over them.
[center]
The discoveries from study of rhythms of activity and eclosion of fruit flies led to the discovery of several ‘clock’ genes and thus our current understanding of the underlying molecular mechanisms that generate a clock.
Objective: To examine / observe whether there is any rhythmicity in activity of fruit flies in our surroundings (home or lab)
- Take slices of any fruit, for example banana or tomato (fresh\rotting) on a piece of paper or any container. For replication, make at least 5 replicate baits of relatively uniform quality or degree of ripeness.
- Set this up on a shelf or counter with ease of observation but with minimal disturbance due to movement of individuals or strong wind currents. Ideally the room must be at temperatures around 25 deg C. Very high or low temperatures can discourage flies. High humidity conditions are also favourable for flies.
[left][floatl]
[/floatl][/left]4.Without disturbing the same, make note of the number of flies visiting the bait at two hour intervals. Take photographs/videos as evidence, using the mobile camera.
5. And is there any pattern repeating? Let’s see!
6. An example of a data spreadsheet for one day is given below. Make such sheets for each consecutive day.
7. Plot a graph of the number of fruit flies observed against time for each day or averaged across days to find out at what time we get the maximum number of fruit flies.
Questions for self-study and interpretation:
- At what time do you see a peak in the number of flies on the bait? When do you see the trough?
- Do you see a recurring pattern in the time of day when maximum flies are spotted on the bait?
- Is this consistent across baits?
- In the assay you performed, did this pattern get affected by any environmental variables or weather conditions in the location of the experiment?
- What might be the ecological significance of the timing of the peak in flies on baits
- What can you infer about the source of the rhythm? How can we find out whether (a) the rhythm is merely a response to the changing light and temperature in the environment OR (b) the rhythm is driven by an internal clock? Do we need to modify this experiment in some way to satisfactorily answer the above question?
4. CULTURING/REARING NATIVE COLLECTED FLIES THROUGH PREPARING HOMEMADE MEDIA.
(Topic: Genetics)
A Simple Approach With Things Available At Home
To achieve our objectives, we started designing a medium with combinations of fruits (like banana and tomatoes).
But then the issue came when we thought of substituting agar (a lab-based solidifying/gelling agent) as we need our medium in a slightly solid condition.
We thought of cornstarch, maida, and rava as alternatives of agar.
All these ingredients were used at different times in combination with fruit.
But then, Rava/Suji/Semolina gave good results.
Good results, in the sense, it gave a perfect consistency as we required.
We wanted to simulate similar kinds of nutrients in the ingredients used in the standard media for fruit flies.
(Note: The Standard Cornmeal Medium is used in laboratories for culturing)
To culture, ingredients of standard Cornmeal media contain nutrients like carbohydrates, sugar, proteins, vitamins, minerals, etc.
In our case, bananas and tomatoes were fruits that provided nutrients like sugar, carbohydrates, proteins.
Rava too has carbohydrates and proteins, but it also solidifies when cooked with adequate water…
Media was improvised every time we made, step by step…
These are the different media prepared during the lockdown period in March-April 2020.
- Banana Rava media
- Tomato Rava Sugar media
- Tomato pieces and Banana peels
- Tomato Rava Sugar & Vinegar
- Tomato Rava Sugar & Yeast Granules
- Tomato Rava Sugar & Coconut oil
- Tomato Rava Sugar & Lemon juice
Our Chosen Home-made Medium to Culture Fruit Flies:
TRSV media (Tomato Rava Sugar and Vinegar media)
Ingredients needed to make ~100ml media. (~25ml media in each of the 4 - 500ml bottles)
• 1 egg-size Tomato
• 2 tbsp Rava/Suji Semolina
• 1 tbsp Sugar
• Vinegar (~5ml in ~30ml media)
• ~250ml Water
- Take a pan.
- Add ~250ml water and allow it to boil.
- Then add 2 tbsp of Rava.
- Add one egg-size tomato chopped.
- Add 1 tablespoon sugar.
- Mix well and cook it on a low flame for 10 minutes.
- Cook till you are sure that all the components of the media are completely homogenized.
- Transfer the medium into clean and dry bottles filling up to approximately 2cm each.
- Add 5ml of vinegar in each media bottle and let it rest.
- Transfer the flies to these media bottles the next day.
[center]
[/center]5. IDENTIFICATION and COMPARISON OF THE NATIVE FLY WITH THE STANDARD LAB BRED DROSOPHILA MELANOGASTER
(Topic: Field biology/genetics)
[center]
[/center]Seymour Benzer, a scientist, had trapped fruit flies from Canton, the USA in the year 1950s.
He made media bottles and transferred the flies into these media bottles.
He put one gravid fly (pregnant female fly) into a bottle. This gravid fly laid eggs, which developed to larvae, pupae, and ultimately adult flies. In this way, similar such bottles of flies were made. These bottles are single line cultures.
These single line cultures were named as CsBz flies (Canton special Benzer flies). The name is given CsBz as these fruit flies are from a place Canton, the USA trapped by Benzer.
So, C stands for Canton, s stands for special and Bz stands for Benzer. Hence CsBz. CsBz flies are identified as Drosophila melanogaster flies.
These flies have been maintained in such bottles from the year 1950s.
Its been 70-80 years and its generations are still being cultured in these media bottles.
[center]
[/center] [center]Figure: Culture bottles of CsBz and Native flies[/center]At the same time, there are generations of fruit flies (from the 1950s) belonging to D. melanogaster species in the native environment now. We can trap these flies, get their generations inside media bottles as Seymour Benzer did, make single line cultures, identify them as D. melanogaster species (if so).
Now, we can have a comparison of the CsBz flies (which is known as the standard D. melanogaster culture) with the native D. melanogaster flies and look for the difference in some of their characters or behaviors like their olfaction, geotaxis, phototaxis, their circadian rhythm, and several other characters.
Protocol for identification of fruit flies.
[center]A Falcon tube with dead flies ![]() [/center] [center]
[/center] [center]
How to get these dead fruit flies?
Method - I
![]() Keep a cotton plug ready of which its one end is dipped in either ethanol, ethyl acetate or chloroform
Keep a cotton plug ready of which its one end is dipped in either ethanol, ethyl acetate or chloroform
![]() Take out some flies from the fruit fly culture bottle into this empty falcon tube
Take out some flies from the fruit fly culture bottle into this empty falcon tube
![]() Close this falcon tube with a cotton plug which was kept ready earlier
Close this falcon tube with a cotton plug which was kept ready earlier
![]() Within 5 mins you will get dead flies in the falcon tube
Within 5 mins you will get dead flies in the falcon tube
Method - II
![]() Take an empty plastic container or a bottle
Take an empty plastic container or a bottle
![]() Take out some flies from the fruit fly culture bottle into this empty plastic bottle or container
Take out some flies from the fruit fly culture bottle into this empty plastic bottle or container
![]() Close the culture bottle
Close the culture bottle
![]() And close the plastic container or the bottle as well with its lid or cap.
And close the plastic container or the bottle as well with its lid or cap.
![]() The flies will be trapped inside this plastic bottle/container.
The flies will be trapped inside this plastic bottle/container.
![]() The flies die within some time due to a lack of oxygen.
The flies die within some time due to a lack of oxygen.
Once we get the dead flies, we can use them for observing and identifying the characteristic features of Drosophilidae family, genus and species.:
Features of identification for Drosophilidae family
Firstly we go about knowing the taxonomic classification-
Kingdom: Animalia, Phylum: Arthropoda, Class: Insecta, Order: Diptera, Family: Drosophilidae
The fruit flies belong to the order Diptera which has unique features of identification:
- One pair of wings (forewings)
- Hindwings reduced to club-like halteres
- A large and moveable head
- Compound eyes that are often very large
- Sucking, piercing and sucking or sponge-like mouthparts (all adapted for a liquid diet)
- The mesothorax (middle segment of the thorax or mid-body) is enlarged, with the prothorax and metathorax small
- Complete metamorphosis, with larvae (maggots) that are always legless, with chewing mouthparts or mouth-hooks, and that often pupate within a hardened case (puparium)
6. UNDERSTANDING THE BEHAVIORAL BIOLOGY OF GEOTAXIS IN ADULT FRUIT FIES.
(Topic: Behavioral Biology)
Protocol
To find out whether your fruit fly culture exhibits a particular type of Geotaxis (movement of a fly against or towards gravity)
- Take your fruit fly culture bottle
- Tap the bottle gently on a soft surface (twice) till we see all fruit flies at the bottom
- Allow the flies to move.
- Record your observation for 10 seconds
Observations to be made
- If fruit flies are seen to move upwards (against the gravity) they are termed as Negatively Geotactic flies
(Which is a normal behavior of fruit flies) - If fruit flies don’t move up, they are termed as positively geotactic flies! (Positive geotactic will indicate that they are towards the ground).
The negative geotactic behavior is the innate escape mechanism exhibited by a Drosophila fly. CsBz flies (Drosophila melanogaster) which is cultured in labs in Corn meal agar media bottles are often seen near the cotton plug at the mouth of the culture bottles. Fruit fly single lines isolated from the environment can be checked for their geotactic behavior and compared with CsBz flies using the Geotactic assay.
The climbing assay is also referred to as the negative geotaxis assay which is used to measure the locomotor behavior of a fly. Flies move against gravity when the vessel containing the fly is tapped on a soft surface and the climbing ability is checked in distance as a parameter. The process involves the segregation of flies into separate test tubes and performing the climbing assay. The materials and methods for the same are as follows :
REQUIREMENTS - drosophila cultures, sugar tube, test tubes, petri dish, paintbrushes, banger, refrigerator, cotton plugs, test tube stand, climbing assay tube (glass tube), marker, stopwatch, logbook.
Protocol for the assay:
- From the selected media bottle consisting of matured or adult flies, transfer the flies into a sugar tube by tapping it on a soft surface and cotton plug the tube.
30cm grduated tube for the Geotaxis assay - Subject the tube to the freezing temperature of -4 degree C in a freezer for 10 minutes.
- Transfer the unconscious flies to a Petri plate.
- With the help of a brush, transfer one single fly to an individual test tube.
- Mark the glass tube with a scale of 30 centimeters.
- Let the flies stay in the tubes and gain proper consciousness for 30 minutes. Insert one single fly from the test tube into the clean, long glass tube and cotton plug the opening of the tube.
- Tap the glass tube on the soft surface so the fly falls on the cotton plug and start the stopwatch as soon as the fly starts climbing.
- Allow the fly to climb for 30 seconds and note the observations.
- Repeat the assay with a single fly for accurate results. (example: perform 3 runs with individual fly).
- The distance traveled by the fly is noted in the logbook and videos are taken for records.
Data represented from the study/observation-
| Fly no. | gender/ sex | Distance 1 (cm) | Distance 2 (cm) | Distance 3 (cm) | Remarks |
|---|---|---|---|---|---|
| 1 | F | 27 | 28 | 28 | Climbs in 20 sec |
| 2 | M | 29 | 29 | 29 | Climbs in 16 sec |
| 3 | F | 25 | 26 | 26 | Climbs slowly in 30 sec |
| 4 | F | 20 | 19 | 19 | Climbs but falls back on cotton |
| 5 | M | 29 | 28 | 29 | Climbs in 18 sec |
| 6 | M | 29 | 28 | 28 | Climbs in 20 sec |
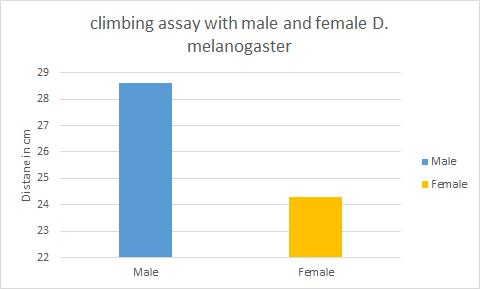
The graph is a sample data which here concludes that both the sexes show negative geotaxis behavior perhaps males show a rapid response when compared to females.
Similarly, the assay can be performed with staged flies (flies of different ages) to
- Check the behavior in mutant flies of same species
- Compare geotaxis between different species
- Use staged flies (flies of different age) to understand the geotaxis behavior of flies with progression in their age.
7. GOOF-UPS IN SCIENCE IS ALSO A FUN WAY TO LEARN.
So, let’s discuss some of them!
Mistakes lead to learning. Here are some mistakes made by CUBists. CAUTION!!
Below are the pointers which highlight our mistakes:
![]() No replicates of the Traps and Culture bottles.
No replicates of the Traps and Culture bottles.
When we Keep Traps for Fruit flies, once we kept only 1 trap and we didn’t get fruit flies in that trap. Our learning was that we should at least keep two traps at a preferred location. Similarly, when we make media, we shouldn’t make just one media bottle, but many of them(at least 3) so that if one media bottle doesn’t work well, we have others to rely on. Also, more media bottles mean more data to interpret what went wrong!
![]() The trap wasn’t kept at the right place.
The trap wasn’t kept at the right place.
Kitchen garbage is a preferable place as it gives out a rotten smell that tends to attract more fruit flies.
![]() Made media for TRAPPING flies!
Made media for TRAPPING flies!
You just need to keep bait (rotten fruits/vegetables) for trapping flies. Media is for culturing flies and not for trapping them.
![]() Media was not cooked properly.
Media was not cooked properly.
The cooking of the medium should be proper which can be done by making sure that all the components of the medium are homogenized (mixed) properly and cooking medium to have the right consistency (semi-solid) until it is transferred to bottles to solidify.
![]() Invitation to contamination!
Invitation to contamination!
What went wrong was that we kept an uncooked raw tomato slice on a cooked media (that was to be kept for maintaining and culturing the flies). So, that uncooked or raw tomato gave rise to contamination in the cooked media.
Note: Do not mix the cooked and raw components arbitrarily.
![]() Lack of anticipation!
Lack of anticipation!
Have an expectation to observe something every day in your traps or culture bottles.
We can expect different stages of the life cycle of fruit flies in our traps and culture bottles every day.
![]() Transferred the media to bottles using a spoon instead of pouring.
Transferred the media to bottles using a spoon instead of pouring.
Using a spoon will cause the medium in the bottles to have an uneven surface.
The prepared medium should have a pouring consistency while transferring into clean and dry bottles.
![]() Lack of clear understanding without proper references.
Lack of clear understanding without proper references.
Referencing gives us a clear, better picture of the work which we are doing. References include the work done on the topic and they can be in the form of research papers or books that are reliable and authentic.
REFERENCES:
- The discoveries of molecular mechanisms for the circadian rhythm: The 2017 Nobel Prize in Physiology or Medicine
- Circadian Rhythms and Sleep in Drosophila melanogaster | Genetics | Oxford Academic
- The discoveries of molecular mechanisms for the circadian rhythm: The 2017 Nobel Prize in Physiology or Medicine - PMC
- Molecular genetics of the fruit-fly circadian clock
- Flies and mosquitoes: Order Diptera - The Australian Museum
Our Collaborators of this great work:
Aashutosh Mule, Anna Malai, Apoorva Satpute, Arunan MC, Aswathy Suresh, Deepika Iyyangar, Drishtant Maruti Kawale, Ganesh C Baskey, Harshita Bhanushali, Isha Pawle, Kavya Honnavad, Kiran Yadav, Kshipra Biresh, Lydia Mathew, Man Masih Beck, Mayur Gaikwad, Nagarjuna G, P. Chitralekha, Rahul Kushwaha, Ruchi Modgekar, Saida Sayyed, Savithri Singh, Shalu Sinha, Yamal Gupta, Yash Sheregare









