Gossip between @Theertha, @sakshiconsultant2002, @Himanshu, @Misbah and @Smiti on Dhanraj’s workshop at Ratnam college on 8 July 2022.
This Gossip started with the hydra and moina that’s been cultured at N.E.S Ratnam College.
-
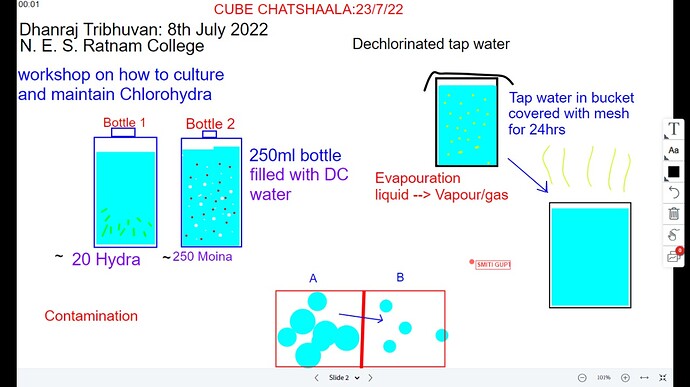
So on 8th July Dhanraj Tribhuvan (Elphinstone college, Mumbai) passed on some Hydras and moinas to Students of N.E.S Ratnam College and mentored them about culturing and maintenance of Hydra.
-
So we talked about how dhanraj explained about his maintenance of hydra and how would we conduct a workshop, also follow it up later on… So here, for that Dhanraj used tap water in a bucket and kept it for dechlorination and used it for culturing both hydra and moina.
-
For culture he took 250ml bottle, in that he filled the D.C (Dechlorinated) water and put the hydra in one bottle and moina in other bottle.
The total number of hydras he gave to Ratnam group was approximately 20 and total number of moina’s were approximately 250 -
After that for showing them the hydra Dhanraj made the bottle half by cutting it in half and then showed us in microscope, i guess that time some of the hydras were misplaced that’s why the number of hydras decreased.
-
Then again we discussed about dechlorinated water-
How do we dechlorinate water?
So we do it be by keep water in a container and covering the container with a cloth. By doing so chlorine in water will evaporate and we’ll get dechlorinated water. -
But there was some confusion -
Does chlorine evaporate or dissolve??
Evaporation is when substances convert from liquid state to gaseous state.
And diffusion is when particles move from higher concentration to lower concentration.
This lead us towards the discussion on diffusion and then osmosis.
Diffusion and osmosis let’s take a look
-
@Misbah tried to explain diffusion ,as we know this is the movement from higher concentration to lower concentration!
-
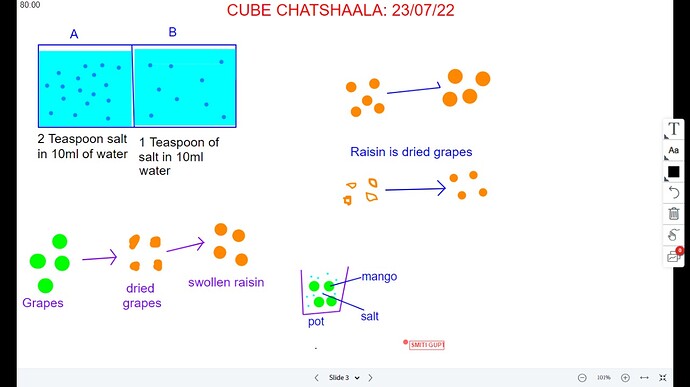
But that statement was not as simple as it looks, so to explain diffusion @Misbah made a line on white board and told on 1 side of line the water concentration is high and other side it’s low (shown with big blue dots and small blue dots)
-
And while giving summary @Theertha told that water is there in beaker and in middle of beaker we have glass plate separating the water concentration… Ik what you’re thinking
-
Then @Arunan Sir CUBE sir started questioning and we got to know that the line in middle would be a semi permeable membrane that will allow the the exchange of water
-
But what about water, does just water (distilled water) will have any concentration? So no! As @Theertha told 15ml & 50ml of water concentration, it wasn’t the concentration but volume. So we can have concentration of salt or suger in water (salt/sucrose solution)
-
I had a question that what can be the example of semi permeable membrane so i took example of raisin but didn’t say it out loud still sir heard and i was like no it’s osmosis
-
But then we discussed and @Chitralekha ma’am told that it’s diffusion and not osmosis
-
So how do we know that it’s diffusion?
For this we discussed what’s raisin it’s dried grapes and we dry it so much that the wall become porous and after removing swollen raisin from water, the raisin gives out water again -
Then we come across the common home lab experiment done by parents & grand parents. That is the "Preservation of raw mangoes in salt"
-
For that, take a glass jar with raw mango and adding salt into it. Later we can see the water content at the bottom. So the water in the raw mango comes outside of the mango through the cell wall!!!
-
Do this cell wall act as a semipermeable membrane. At the same time salt in the jar goes inside to the raw mango !!
-
So can we call Preservation of raw mango in salt as an example of Osmosis (Movement from higher concentration to lower concentration through semipermeable membrane!!!