CUBE ChatShaala Summary – 30 July 2025
CUBE ChatShaala Summary – 30 July 2025
 Focus of Today’s Discussion
Focus of Today’s Discussion
Building on Tarannum’s ongoing investigation into making curd without a starter culture, today’s ChatShaala delved into whether individual amino acids like glycine have any direct role in curd formation—or whether the real mechanism lies with acidification and protein chemistry.
 Experimental Model Recap
Experimental Model Recap
-
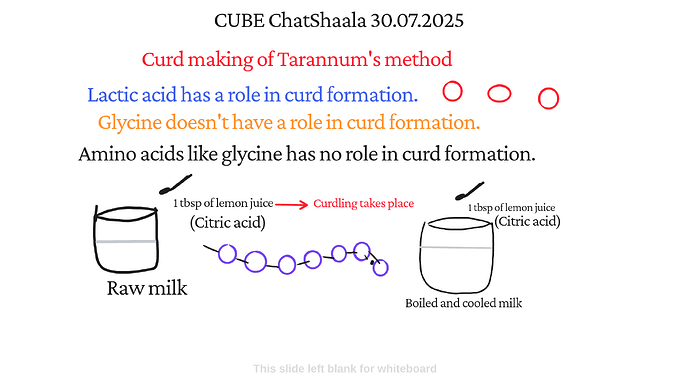
Two milk conditions were discussed:
-
Raw milk + 1 tbsp lemon juice (citric acid)
Boiled & cooled milk + 1 tbsp lemon juice
In both setups, curdling occurred, indicating that acidification alone—regardless of prior boiling—was sufficient to cause protein coagulation.
 Key Whiteboard Insights
Key Whiteboard Insights
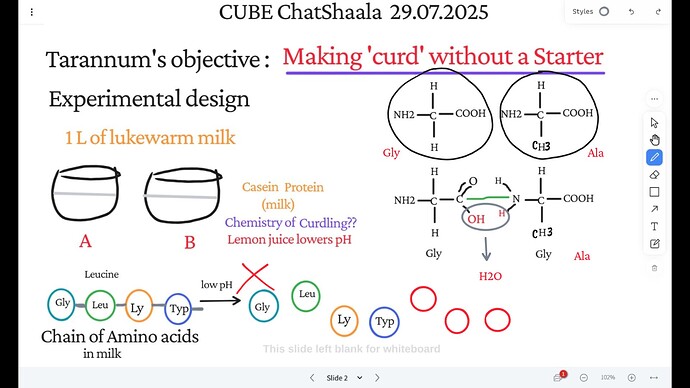
Glycine, a simple amino acid, was shown as a repeating component in the casein protein chain.
The diagrams clarified that glycine (and similar amino acids) play no direct role in curdling; instead, the structure of the casein micelle is altered by a drop in pH, not by individual amino acids.
The real agent is the citric acid in lemon juice, which lowers the milk’s pH, reaching the isoelectric point of casein (~pH 4.6). This neutralizes charges on micelles, making them stick together and separate from the liquid (whey).
 From Protein Chains to Curd Blobs
From Protein Chains to Curd Blobs
At low pH:
The normally stable micelles of casein lose their negative charges.
This removes electrostatic repulsion, leading to aggregation of the proteins.
The result: visible clumps of curd form—without any need for starter bacteria, rennet, or enzymes.
This process is chemical, not microbial—though in traditional curd-making, microbes (like Lactobacillus) play the acid-producing role.
 Final Conclusion from Today
Final Conclusion from Today
Lactic acid (whether added directly via lemon juice or produced by bacteria) is the key agent in curd formation.
Glycine or other individual amino acids are irrelevant to the process of curdling itself.
 Provocative Questions for General Engagement
Provocative Questions for General Engagement
-
If lactic acid is enough to form curd, is the starter curd even necessary?
-
Can raw milk and boiled milk both form curd with lemon juice—but taste differently?
-
What happens to vitamins or fats during this acidic transformation? Do they also separate?
-
Would vinegar (acetic acid) or tamarind (tartaric acid) do the same job as lemon juice?
-
Is curd made without bacteria really the same as traditional curd—or just chemically identical?
-
If glycine doesn’t matter—what’s actually inside the curd? What does it trap or release?
 What I Personally Learned Today
What I Personally Learned Today
Today’s ChatShaala made a clear distinction between protein structure and protein function. I often imagined that amino acids like glycine might actively “cause” curdling. But now I understand that curdling is driven by environmental change (pH drop), not intrinsic amino acid behavior. This shift in perspective helps clarify how simple kitchen chemistry mirrors complex molecular processes—and challenges some cultural assumptions about how curd “must” be made.
I also found it inspiring how we connected food, chemistry, and molecular biology in a way that empowers anyone to replicate and test ideas at home. That’s science at its most accessible and democratic.
 Reference
Reference
@Arunan @sakshiconsultant2002 @sneha
@Chitralekha @KiranKalakotiR @akanksha and others.