CUBE Chatshaala Meeting Summary : (16/01/2026)
CUBE Chatshaala Meeting Summary : (16/01/2026)
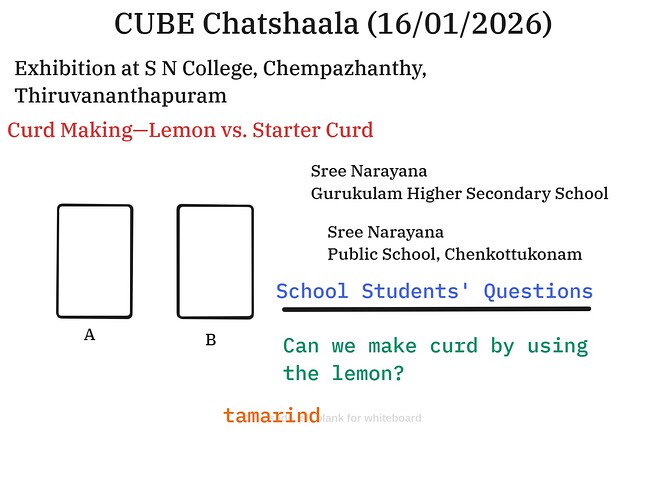
The session began with Sailekshmi’s CUBE homelab experiment at the exhibition at S N College, Chempazhanthy, Thiruvananthapuram, which was a lively exploration of biological and chemical processes, specifically the transformation of milk into curd. The discussion was sparked by a fundamental question posed by students from Sree Narayana Gurukulam Higher Secondary School and Sree Narayana Public School, Chenkottukonam: “Can we make curd by using lemon?” Most of the school students mention using tamarind to make curd.
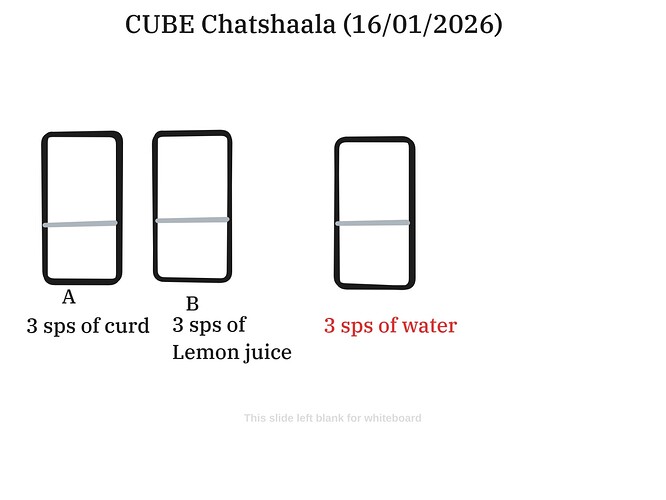
To address this, the participants developed a comparative experimental design. Three separate beakers were used to observe the reaction of milk to various additives:
- Beaker A: Milk with 3 spoonfuls of starter curd (for biological control).
- Beaker B: Milk with 3 spoonfuls of lemon juice (acid intervention).
Simti Gupta’s suggestions:
- Control Beaker: Milk and 3 spoonfuls of water (neutral control).
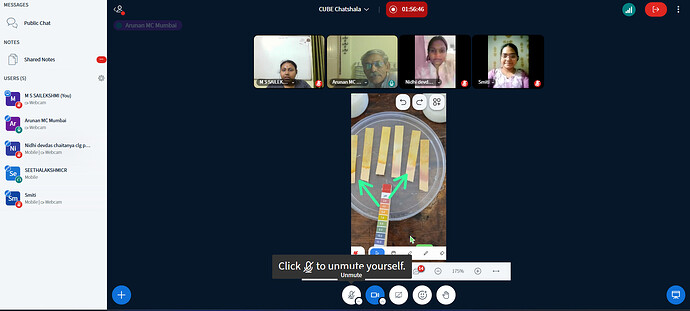
The discussion focused on the “Science of Souring.” While lemon juice (citric acid) can coagulate milk proteins (casein) through a rapid pH drop, the resulting product differs structurally and microbiologically from curd produced by Lactobacillus in a starter culture. The use of pH strips, as demonstrated in the experimental data, provided visual confirmation of the acidity levels required to cause protein denaturation. This experiment provides an accessible entry point for students to understand that “curd-making” is more than just a kitchen chore, but a sophisticated interplay of pH levels and microbial fermentation.
 Queries for the CUBE Community
Queries for the CUBE Community
-
If lemon juice makes milk thick and sour, why don’t we call it “lemon curd” for our breakfast? What is the fundamental difference between “acid-curdled milk” and “microbially fermented curd” in terms of nutrition and shelf life?
-
The whiteboard mentions tamarind. If we replace lemon juice with tamarind pulp, would the protein structure change differently? Does the type of acid (citric vs. tartaric) change the texture of the resulting curd?
-
Looking at our pH strips, we see a shift toward the red spectrum. At exactly what pH value does milk stop being a liquid and start becoming a solid? Is this threshold the same for cow milk, goat milk, and plant-based milks?
 The TINKE Moments (This I Never Knew Earlier)
The TINKE Moments (This I Never Knew Earlier)
-
Students actively manipulated common kitchen ingredients (lemon, curd, water) to observe a chemical change, moving science from the textbook to the beaker.
-
The investigation was driven by a student-led query, proving that the best science starts with a “What if?”
-
Participants noticed the color change on the pH strips, correlating the visual shift (yellow to orange/red) with the physical thickening of the milk.
-
A realization emerged that lowering the pH is the common denominator in both lemon-added milk and starter-added milk.
-
The group formulated an explanation that while acid (lemon) mimics the result of fermentation, it does not replicate the process of bacterial growth.
 Gaps and Misconceptions
Gaps and Misconceptions
-
The “Same Result” Fallacy: A primary misconception among students was that any sour agent creates the same curd. There is a significant gap in understanding the difference between acid coagulation (instant) and bacterial fermentation (slow). One is a chemical reaction; the other is a biological life cycle.
-
pH vs. Probiotics: Students often assume “sour” equals “healthy.” The gap lies in realizing that lemon-curdled milk lacks the live Lactobacillus cultures (probiotics) found in traditional curd, which are essential for gut health.
-
The Role of Temperature: While the experiment focused on additives, the role of ambient temperature in facilitating bacterial growth in Beaker A versus the chemical reaction in Beaker B was not fully explored.