CUBE ChatShaala | 24 December 2025
Meeting Summary
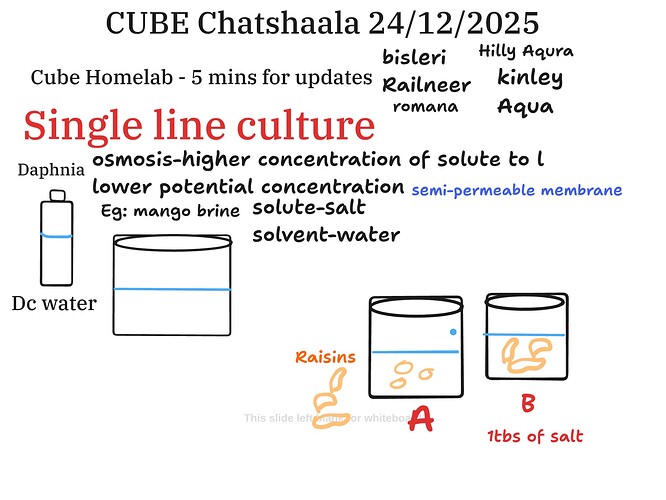
Today’s CUBE ChatShaala opened with quick homelab updates and smoothly transitioned into a focused discussion on single-line culture, using Daphnia and simple everyday materials to explain a fundamental biological principle—osmosis.
The whiteboard discussion clearly illustrated how water moves across a semi-permeable membrane from a region of lower solute concentration to higher solute concentration. Familiar examples, such as mango brine, helped ground the concept: salt as the solute and water as the solvent.
A practical demonstration using raisins in two containers (A: plain water, B: water with one tablespoon of salt) highlighted osmotic behavior. In plain water, raisins swelled due to endosmosis, while in salt solution they shrank, demonstrating exosmosis. This simple setup reinforced how concentration gradients influence cellular and organismal responses, linking classroom theory with observable outcomes.
The session emphasized how low-cost experiments can effectively communicate complex biological ideas and encourage scientific thinking beyond textbooks.
 What I Learned Today
What I Learned Today
-
Complex biological processes can be explained powerfully using simple, relatable experiments.
-
Osmosis is not just a textbook definition but a living process visible in everyday materials.
-
Careful control of variables (solute concentration) is key to meaningful scientific observation.
-
Teaching science becomes impactful when learners are encouraged to predict, observe, and reflect.
 TINKE Moments (This I Never Knew Earlier)
TINKE Moments (This I Never Knew Earlier)
-
Raisins act as excellent natural models for demonstrating osmotic movement.
-
Even small changes in salt concentration can cause visibly different biological responses.
-
Concepts like “water potential” become intuitive when seen rather than memorized.
-
Homelab-style demonstrations can be as effective as formal laboratory setups.
Gaps and Misconceptions That Emerged
-
Osmosis was initially confused with simple diffusion, ignoring the role of a semi-permeable membrane.
-
Some participants assumed water always moves towards salt, without considering relative concentrations.
-
The idea of “higher” and “lower” water potential needed repeated clarification.
-
There was a tendency to memorize definitions rather than reason through the process.
 Photographs during Chatshaala
Photographs during Chatshaala
 Provocative questions of the discussion
Provocative questions of the discussion
- How would temperature changes affect the rate of osmosis in the same setup?
- Can similar experiments be designed to explain plasmolysis in plant cells?
- How does osmotic balance maintain survival in freshwater organisms like Daphnia?
- What other household materials can model biological membranes effectively?