Highlights of Context to Curriculum whatsapp group:November 2022
Sakshi @sakshiconsultant2002 ,Maharashtra
Theertha,Homelab Cherukkad,Kozhikode
@KiranKalakotiR ,Moradabad,Uttarpredesh
@Shraddha276 Panvel ,Maharashtra
Cardamine :A Close relative of Arabidopsis .
Cardamine is considered as a close relative of Arabidopsis .
Easily available.
Shows genetic similarity with Arabidopsis .
Easy to maintain
Short lifecycle within 2 week
Cardamine: Substitute Indian Model System for Arabidopsis
https://metastudio.org/t/cardamine-substitute-indian-model-system-for-
arabidopsis/3663
Disscussion on context to curriculum by @Shraddha276 @Himanshu sir @Arunan sir and others
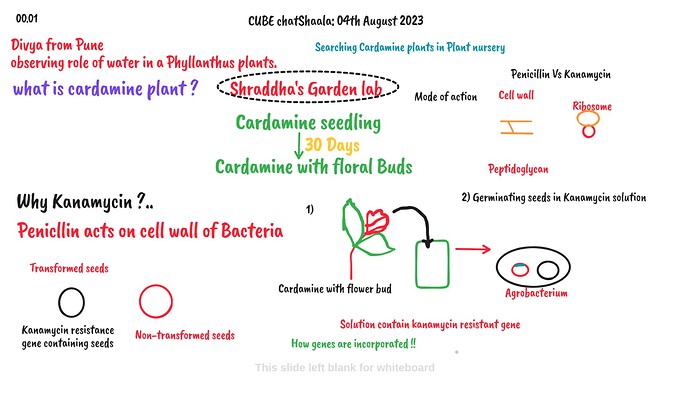
Why do we need to incorporate this kanamycin gene into the plant?
In prokaryotics, 70S ribosomes are present which is made up of 50S and 30S
subunits.Kanamycin is an antibiotic which inhibits small subunit of ribosomes
i.e. 30S subunits in prokaryotics.
But Cardamine is eukaryotic plants so they having 80SRibosomes(60S + 40 S) in
cytoplasm but in mitochondria and chloroplast, because of prokaryotic in origin,
they have 70 S ribosomes similar to prokaryotics.Therefore, we will use
Kanamycin resistance gene as marker gene for selection of transformed and non
transformed seeds.when we incorporate Kanamycin resistance gene by floral dip
method in Cardamine we will get transformed seed - because of Kanamycin
resistance gene present, this seeds can germinate and grow further in culture
having Kanamycin antibiotics. They will be green in colour.
whereas, in same culture non transformed seedlings, they will be in pale green
colour with shortened in length of radical and plumule.
Hence, to distinguish between transform and non transform seedlings we use
Kanamycin resistance gene when we incorporate Kanamycin resistance gene by floral dip method in Cardamine we will get transformed seed - because of Kanamycin resistance gene present, this seeds can germinate and grow further in culture having Kanamycin antibiotics. They will be green in colour. whereas, in same culture non transformed seedlings, they will be in pale green colour with shortened in length of radical and plumule. Hence, to distinguish between transform and non transform seedlings we use
Kanamycin resistance gene leaves.
How can we introduce desired kanamycin resistance gene into the
agrobacterium?
To introduced foreign gene into bacterial cell, we can do cloning.
In cloning, by using restriction enzymes we can insert gene of interest into a
bacterial plasmid.
Ranchi CUBist was performed experiment by using
1.Agrobacterium Strain- LBA4404
2 Vector- pCAMBIA 2301
3.They were culture Agrobacterium in LB Broth with kanamycin and rifamycin
antibiotics.
How can we transfer a gene from bacteria to plants?
There are different bacteria which contain chromosomal DNA and extra
chromosomal DNA in their cytoplasm.
Extra chromosomal DNA is known as plasmid. Bacteria can transfer copy of
plasmid to another bacteria (which do not have plasmid in their cytoplasm) . This
process take place by physical contact in bacteria with help of pili. This is
conjugation.
Why? How? Agrobacterium is helping us?
Because it’s soil bacteria which attracted (when plant get wounds like any cut,that
time dicote plants released phenolic compound.Acetosyringone)
and infect. It causes crown gall disease by formation of tumor.
During infection, bacteria transfer T-DNA which contain some genes. This DNA
contain some genes which help bacteria to survive within plant cells.
For transformation of gene from bacteria to plant cells, Agrobacterium use their
plasmid which is Ti-plasmid(tumor inducing plasmid).
This Ti plasmid contain T-DNA within restricted borders so it can cut and
transferred,vir genes, ori site.
During transformation, many virulence genes are present on plasmid (but outside
T-DNA )which get activated and form passage between bacterial cell and plant
cells and help in transformation of T-DNA.
If we cloned our gene of interest in between borders of T-DNA region then our
gene of interest can transform into plant genome.
Conventionally,genetic engineering performed by plant tissue culture.
plant tissue culture is laboratory method for gene transfer in which plant cells
grow in aseptic conditions by supplying nutrients, Agrobacterium, and gene of
interest in the media itself. And then similar mechanism use here for
transformation of bacterial gene to plants.
For performing this method, sterile conditions must be there. So you should have
separate space, for maintaining cells you will require MS media, also required
skill person to perform this experiment.
It is technique through which we can incorporate the gene of interest by simply
dipping the floral buds of Cardamine in Agrobacterium solution containing gene
of interest. Due to simplicity in performing, Agrobacterium mediated
transformation by floral dip method.
Let’s see how we can proceed to achieve
GM Cardamine plant at your home lab.
- Initially, culture Cardamine plants in small pots containing soil till 4th
compound leaf stage.
2.after 4th compound leaf,there are possibility of Floral inflorescence will
come(according to earlier data collected by me in CUBE HBCSE)
Once floral bud start initiates to till formation of floral bud it’s required 5-6 days.
Till that time we can start Culturing Agrobacterium with kanamycin resistance
gene on LB Broth.
Then we can resuspened cell in 5% sugar (for maintaining moisture) & silwett
L- 77 solution (as surfactant) on day of performing floral dip experiment.
Then, we will dip inflorescence (which is in bud conditions) in Agrobacterium
cell suspension solution for few seconds.
Then we will cover plant with help of plastic cover for 16- 24 hrs to maintain
moisture and for avoidance of fungal contamination from outside environment.
Then, by maintaining moist soil with water, we will allow plant to grow. Once
seed pods get mature it will be in pale yellow colour. In this stage we can collect
seeds which can be transformed or non transformed.
What are the morphological changes that would be expected after a successful
gene transfer in cardamine plant?
our gene of interest is kanamycin resistance gene. So transformed seeds can
germinate and grow well in kanamycin containing media.
It’s act as marker gene. Therefore,Role of Kanamycin resistance gene is only
inhibits effect of kanamycin on seeds so, it will not change morphology of Cardamine plants.
@Shraddha276 @KiranKalakotiR @sakshiconsultant2002 @Rechel_tirkey and others