Hey Everyone, This is Drishtant Maruti Kawale [F.Y. B.Sc. (H) Biotech] from CUBE Elphinstone College, Mumbai.
I am working on the model organism Moina. Moinas can be studied as a model organism because first of all, it is easy to culture, gives more offsprings in less time (parthenogenetically) /short life cycle due to which gene expression can be studied in less time, it is sensitive to changes in water chemistry.
So, the future scope is to study epigenetics of Moina in detail, first understand the process on the observational basis rather than the experimental one. This is an interesting field of study and can be studied on cancer and other knowledgeable fields.
Introduction:
Classification:
Kingdom: Animalia
Phylum: Arthropoda
Subphylum: Crustacea
Class: Branchiopoda
Order: Cladocera
Family: Moinidae
Genus: Moina
Objectives:
Immediate: Culturing of moinas and to maintain a red Moina culture.
Short term: To check for colour change in Moinas after a week or so.
Long term: To study epigenetics (the study of inheritable phenotype without altering the DNA sequence) and specifically DNA acetylation in Moinas using HDAC inhibitor Valproic acid (VPA) and to study the effect of unfavourable conditions like hypoxia in Moina and the probable reasons for the responses (changing to red colour) shown by Moinas in such conditions.
So, today 3 culture bottles L1, L2, L3 were prepared at CUBE HBCSE, Mumbai. The Moinas in these bottles were taken from the existing cultures at HBCSE.

L1 & L2: 750ml DC Water + 5 colourless Moinas + 3 drops of milk (1 drop - in 250ml DC so 3 drops in 750ml)
L3: 250ml DC Water+ 5 colourless Moinas + 4 drops of milk (Hypoxia)
Tomorrow’s Expectations: 5-7 Moinas in all bottles
We are performing the VPA set up so we will need more culture bottles, (in fact more red moinas because we will check the effect of the HDAC inhibitor on Red Moinas which would take more days to become colourless at various concentrations of VPA) which will be prepared in the coming days.
I know that more culture bottles could have been prepared today itself!
We’ll keep posting literature regarding our set up along with the references.
What is VPA?
These are medications primarily used to treat epilepsy and bipolar disorder and to prevent migraine headaches.
Why are we using it on Moinas or as an HDAC inhibitor?
Well-tolerated antiepileptic drug valproic acid is a powerful HDAC inhibitor. Valproic acid relieves HDAC-dependent transcriptional repression and causes hyperacetylation of histones in cultured cells and in vivo. Valproic acid inhibits HDAC activity in vitro, most probably by binding to the catalytic centre of HDACs. Most importantly, valproic acid induces differentiation of carcinoma cells, transformed hematopoietic (stem cells which give rise to other blood cells) progenitor (parent) cells and leukemic blasts from acute myeloid leukaemia patients. Moreover, tumour growth and metastasis formation are significantly reduced in animal experiments. Therefore, valproic acid might serve as an effective drug for cancer therapy.
Reference for VPA as an HDAC inhibitor: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC125788/#targetText=Valproic%20acid%20relieves%20HDAC-dependent,the%20catalytic%20center%20of%20HDACs.&targetText=Therefore%2C%20valproic%20acid%20might%20serve,effective%20drug%20for%20cancer%20therapy.
So, the question may arise that why to study Haemoglobin of Moina?
Haemoglobin in Moina:
Haemoglobin is a protein which carries oxygen (1 Hb molecule carries 4 Oxygen atoms) to all parts of the body in humans as well in Freshwater Moina s. It is present throughout the bloodstream in the body in humans and it’s level increases or decreases as per the requirement of the organism. But, in moinas, Haemoglobin expression can be seen in a span of few days by altering their environmental conditions like Normoxia (Normal Oxygen concentration in their environment i.e. water) and Hypoxia (Less Oxygen concentration in their environment). As a result, the moinas turn pale yellow to red in colour after keeping them in hypoxic condition because, as there is less oxygen already, more Hb would be required by the organism to fulfil its bodily functions as it is the carrier of oxygen. It restores back to colourless or white colour after keeping it back in the normoxic condition.
Literature:
In normoxic conditions i.e. When there is normal dissolved oxygen concentration, the moinas are colourless, but a change in colour is observed after some days when they are kept under hypoxic (low dissolved oxygen) conditions, their colour changes from colourless to pale yellow and then to red. They tend to develop more or extra haemoglobin to increase the uptake of oxygen from the water. The Hb uptake varies in response to environmental changes. The form of Hb protein which contains oxygen is Oxy-Haemoglobin and is responsible for the red colour of moinas in hypoxic condition, the quantity of whose depends upon the dissolved oxygen gas concentration in the water. We have observed that the amount of dissolved oxygen is inversely proportional to the amount of Hb produced.
We hypothesise that change in the colour of moinas is due to the change in their gene expression. Of the four Hb genes: dmHb1, dmHb2, dmHb3, dmHb4 (all located on a single chromosome), in the *Moinas *, any two (which two exactly are not known to us) are active i.e. Produce Hb in normoxia and all four are active in hypoxia producing extra Hb, as, for that period of time it is irreversible change and not a mutation, we thought it as to be an epigenetic (phenotypic inheritable change in morphological/observable characteristics without change in DNA sequence) change induced due to change in environmental conditions.
So, when epigenetic changes take place changes in morphology, physiology also takes place but no change in DNA content takes place.
In case of our freshwater crustacean, this occurs during the hypoxia condition where with low oxygen concentration, the Hb concentration increases up to 10 times the normal concentration due to the upregulation of Hb. Upregulation in simple terms means on the action of external stimulus, the cell increases the number of cellular components mainly RNA or protein. Here, Hb is upregulated which is why moinas appear red in colour.
The hypoxic condition stimulates the changes in the body of Moina which leads to histone modifications.
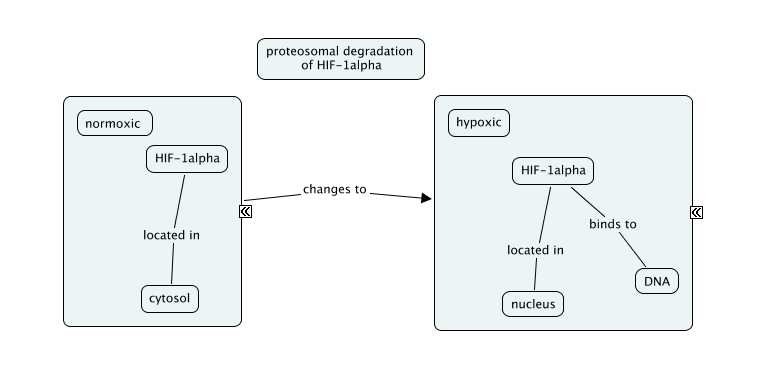
The gene is a part of DNA. In moinas during the hypoxic condition, some enzymes like HAT (histone acetyltransferases) become active which leads to acetylation of histone protein. During the acetylation, the acetyl group CH3COO- removes the positive charge from the histone protein which was a positively charged protein due to the presence of amino acids Lysine and Arginine which are basic in nature due to the presence of –NH2 group in them. Earlier, there was an octamer between histone and DNA which were bound by the nucleosome which is the fundamental unit of DNA packaging but now, as a result of acetylation, there will be no electrostatic force of attraction between DNA and Histone protein due to which DNA becomes loose and hence RNA polymerase with the help of transcription factor, HIF-1-alpha or hypoxia-induced factor 1 alpha reads the DNA strand and transcribes it to RNA and then translates it into Haemoglobin protein which is needed by the moinas. The promoter regions of the globin genes each contain hypoxia response elements (HREs) as potential binding sites for HIFs.
So, after the addition of Valproic acid, the reverse of the above process happens. The HDAC (histone deacetylases) are not able to deacetylate the histone as a result of which the Hb protein will be produced until the concentration of VPA decreases and hence the Moinas will remain red.
Proposed Experimental Setup
Objective: To study the effect of HDAC inhibitor Valproic Acid (VPA) on Red Moinas at different concentrations.
Requirements: Valproic Acid tablet 200mg, test tubes, DC water and milk.
We are going to use 200mg Valproic acid tablets as the HDAC inhibitors. We are going to take different concentrations of the VPA solution – 5ug, 10ug, 50ug, 100ug, 500ug (ug is microgram) and going to check the number of days taken by the HDAC inhibitor to inhibit the deacetylation in the Moinas at the above-mentioned concentrations.
Preparation of Valproic Acid solution as well as its different concentrations:
Take 200mg VPA tablet and dissolve it in 200ml DC water. The solution now is of 1ug/ml.
Now, to prepare 5ug concentration, take 5ml of the above 1ug/ml solution and add it into 995ml of DC water.
Repeat the above step to prepare 10ug, 50ug, 100ug, 500ug concentrations.
Take 12 test tubes and add 10ml of DC water in each of them. To that add 5 red moinas (except in the colourless control one – where 5 colourless moinas would be added) which were obtained by inducing hypoxia.
Now, add the prepared concentrations dropwise in 10 of those test tubes (5 main and 5 duplicate) and the remaining 2 test tubes would be Control tubes – one red control and the other colourless control.
After that the moinas need to be fed with milk in terms of dilutions.
As per the protocol, we add 1 drop of milk in 250ml of DC water, so for 25ml of DC water, we will add 1:10 dilution of milk i.e. 1 drop of milk and 10 drops of water and now for 10ml of DC water, 1:25 dilution.
We hypothesise that as the moinas are kept in different concentrations, they will get colourless in varying time. For example, the 5ug moinas will turn back into colourless in less time as compared to the 50ug ones due to the lesser concentration of VPA. The red moinas which are to be kept in the water containing VPA solution at different concentrations will take more to revert back to colourless at different concentrations because, in the presence of VPA which is an HDAC inhibitor, the DNA deacetylation will take time. This will only take place when gene regulation will happen by DNA acetylation.
This assay would be performed only and only when there are a considerable number of red moinas with us!
References
- NFC: Moina – Russian Red Daphnia, Tuesday 14 September 1999 – Hemsath Gay
- Moina (Crustacea: Anomopoda, Moinidae) in the Czech Republic, November 4, 2002,– Adam Petrusek Department of Hydrobiology, Charles University, Prague, Czech Republic
- The Haemoglobin of Daphnia, 5 August 1947 - H. Munro Fox F.R.S., Belford College, University of London
- HIF at a glance, Journal of Cell Science, 2009 – M. Christiane Brahimi-Horn, Jacques Poussegur
- Hypoxia-induced synthesis of haemoglobin in the crustacean Daphnia magna is hypoxia-inducible factor-dependent, Journal of Biological Chemistry, May 28, 2008, Thomas Gorr Division of Haematology, Department of Medicine, Brigham and Women’s Hospital, Harvard Medical School, Boston, Massachusetts, Yamagata H, Bunn HF, Cahn JD.
- Transcriptional and Translational Relationship in Environmental Stress: Frontiers in Physiology, 02 July 2018 – Jingyi Jia, Lu Li, Xiangjiang Liu, Chengqiang Lei, Ying Dong, Guoqiang Wu and Guangfu Hu – College of Fisheries, Huazhong Agricultural University, Wuhan, China
- Sequencing of Hb Gene 4 (dmhb4) and Southern Blot Analysis provide Evidence of More than Four Members of the Daphnia magna Globin family: Bioscience, Biotechnology and Biochemistry, 22 May 2014 Marc Wolf, Frank Nunes, Ralph Pirow, Alexandra Wendler, Desiree Spiering, Rudifer J. Paul.