[size = 6]Molecule Shapes[/size]
We discussed the concept of “Molecule Shape and Geometry” from the scratch via PhET simulation.
- Link of the simulation: Molecule Shapes (colorado.edu)
- Link of the recording: click here
Reflection
-
Determine the shapes of AB[size = 0.5]x[/size]L[size = 0.5]y[/size] type of molecule where x and y are integer numbers greater than or equal to zero (x is the number of atoms of B and y is the number of lone pairs L) ?
-
How to determine the bond angle for a planner molecule?
-
What will determine the molecular shape: the number of atoms and lone pairs or the number of bonds attached to the central atom?
-
For a AB[size = 0.5]4[/size] type molecule why the geometry is tetrahedral and not square planner?
-
What is the principle in terms of bond angle behind the formation of the shape for a particular molecule shown in the simulation and not any other shape is preferred?
-
What are lone pair and bond pair electrons?
-
How the lone pair is contributing in the differentiation of molecular and electronic geometry?
-
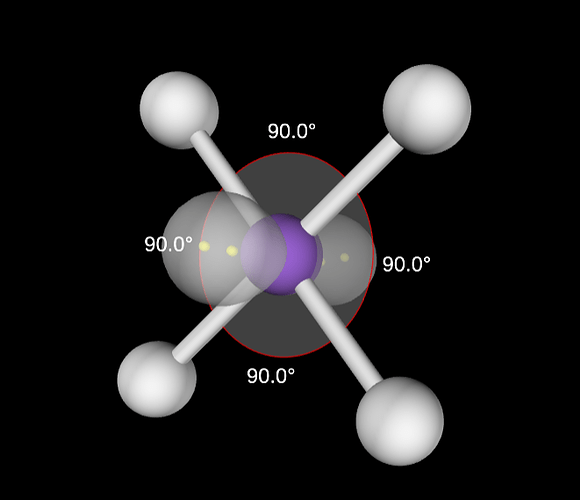
Why we can’t switch one of the bond pair and one of the lone pair electrons for the following molecule though then also we are getting all the bond angels as 90 degrees? What is the problem with the new structure?
-
Share your thoughts, questions and comments regarding the simulation and your experience during exploring the simulation.
Please click here for the part II of this discussion.
We welcome all kind of suggestions and feedback if this was useful and how can be collectively make it better.
Acknowledgements
Discussion
Collaborators in video resource creation: Prof Savita Ladage, Deepak Arora, Hanza George, Mursaleen Shaikh, Ravi Sinha, Ashish Pardeshi, Indrani Das, Muskan Sharma, Swarnava Mitra
Credits
PhET Interactive Simulations
University of Colorado Boulder
https://phet.colorado.edu